Highlights on the 6th International ALPE Congress on Achondroplasia & Other Dysplasias

| Figure 1. ALPE congress main image |
Participants had the opportunity to meet face to face with key experts and many had the opportunity to get an expert medical opinion in specific situations and complex cases, in diverse medical fields.
It was a very extensive congress with thematic sessions crossing from medical and surgical updates, current ongoing research and psycho-social aspects of achondroplasia.
Prof. Sérgio Sousa (member of Beyond Achondroplasia Advisory Board), opened the congress in the first session on "Clinical Genetics of Skeletal Dysplasias", with an introduction on bone formation, the rationale behind the names and designations of specific dysplasias and also presented ERN BOND, the European Reference Network for Rare Bone Diseases and the multidisciplinary clinic for rare bone diseases he coordinates in Portugal. The other two speakers in this session, Joana Bengoa, and Karen Heath discussed the diagnosis and genetic counseling on bone dysplasias.
| Figure 2. Dr. Sérgio Sousa presentation. Credits: Beyond Achondroplasia |
This was followed by the session "Future Challenges in Skeletal Dysplasia" with Prof. Ravi Savarirayan, from Murdoch Children’s Research Institute, Melbourne, addressing the development of medicines for achondroplasia and Dr. Laurence Legeai-Mallet, researcher at Imagine Institute, Descartes University in Paris, explaining the studies on BGJ 398, or Infigratinib, the molecule QED Therapeutics is developing for achondroplasia.
An very interesting presentation during the session "Overview of current research on achondroplasia" was made by Dr. Elvire Gouze, scientific founder of Therachon and head researcher at Valrose Institute of Biology, in Nice, presenting the basic research on TA-46, its effects on obesity in achondroplasia and current research in this field, as well as the results, achieved so far, in pre-clinical studies (general overview of the soluble FGFR3, now TA-46, can be seen here).
On the 2nd day, Dr. Guirish Solanki, Consultant Paediatric Neurosurgeon at the Birmingham Children’s Hospital, Birmingham, did an extraordinary presentation during the session "Multidisciplinary assessment: main issues" on the brain and intracranial changes and neurologic impact of complications that occur in achondroplasia. After this presentation, Dr. Christoph Kampann, Head of the Department of Paediatric Cardiology, Mainz, Germany, approached electrocardiographic (heart) alterations in achondroplasia.
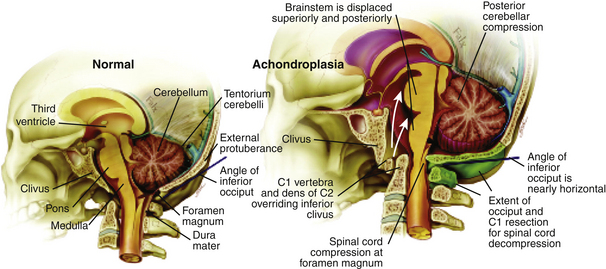
| Figure 3. Normal findings (left) compared with findings in the cervicomedullary junction in patients with achondroplasia (right). The horizontal orientation of the posterior rim of the foramen magnum in achondroplasia causes kinking of the cervicomedullary junction. The area of bone removed during the cervicomedullary decompression is depicted in green (right). Credits: Bagley CA, et al. Cervicomedullary decompression for foramen magnum stenosis in achondroplasia. J Neurosurg. 2006 Mar;104(3 Suppl):166-172. Showed by Dr. Solanki in the ALPE congress |
One of the highlights was the "Advances in clinical research" session in which, 4 of the 5 companies that are currently conducting research on innovative medicine for achondroplasia, presented their work: BioMarin (BMN-111), Therachon (TA-46), Ascendis Pharma (TransCon CNP) and QED Therapeutics (Infigratinib). Ribomic Inc. was absent and the researchers from Nagoya University studying Meclozine for achondroplasia couldn´t attend.
Pharmaceutical companies presentations
Dr. Elena Fisheleva, BioMarin Medical director, explained why companies need to conduct Natural History studies on rare diseases, that are observational studies. BioMarin current study is LIAISE, more details here and where these fit in the research of new drugs. Observational studies will facilitate the understanding of how rare conditions affect patients throughout life and can be used as a comparison for the changes these drugs are causing in the study participants. Fisheleva also presented where BioMarin's studies are taking place in the following infographic:
| Figure 4. BioMarin presentation at the ALPE congress. Credits: BioMarin |
Dr. Kennett Sprogøe, Ascendis Pharma Vice-President of Product Innovation, did a brief introduction on the company pipeline (products in development) and presented a timeline for the next steps the company will take on the development of TransCon CNP, shown below: The predicted timings are the beginning of the Natural history study at end of 2018 and phase 2 in mid 2019.
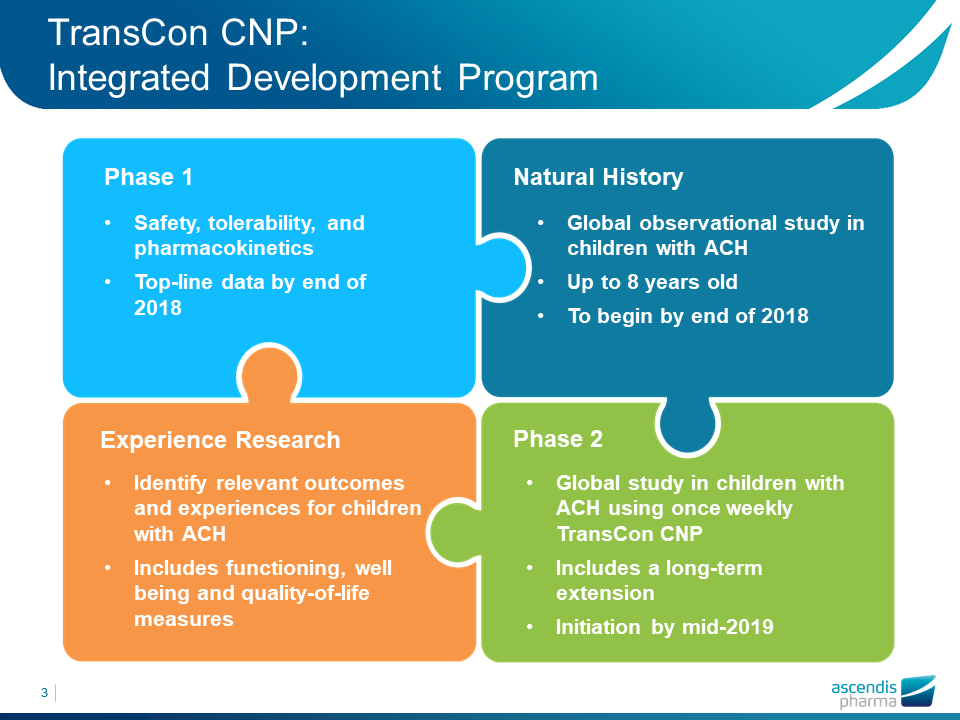
| Figure 5. Ascendis presentation at the ALPE congress. Credits: Ascendis |
Dr. Federico Bolognani, Vice-president Head of Clinical Science of Therachonpresented how the development of TA-46 is progressing in a timeline. The company started the Dreambird study in June 2018, to assess the burden of complications of achondroplasia in approximately 200 children across multiple sites in Europe, Canada, and the United States. This study aims also to support the development of key biomarkers of bone tissue growth. Therachon estimates to conduct phase 2 in 2019 and 2020, aiming to obtain market approval by 2023, which can be seen below:

| Figure 6. Therachon presentation at the ALPE Congress. Translated to English with permission from Therachon. Credits: Therachon. |
Susan Moran, Chief Medical Officer of QED Therapeutics, presented the company to the audience and announced an ongoing preclinical work to find the lowest efficacious and safest dose to start treatment in children. QED plans to open up an observational study in some regions in late 2018 or early 2019 and after that, start the dose-finding phase in some regions in the second half of 2019.

| Figure 7. QED presentation at the ALPE Congress. Credits: QED |
After this, and in a summary mode, the predictions on the development of innovative medicines for achondroplasia are the following:
| Figure 8. Morrys Kaisermann's presentation at the ALPE Congress. Credits: Tratando Acondroplasia |
Patient organizations
The day 2 of the congress ended with the session "International Patient Organizations: building networks", with representatives from 19 countries, and the moments all had for a brief presentation, enlightened the audience on the social intervention patient organizations have and actions that are still needed to be taken towards improving quality of life of people with skeletal dysplasias and their families.

| Figure 9.Patient representatives from 19 countries. Credits: ALPE Foundation |
Overall, the ALPE congress captured what the key points around achondroplasia and other dysplasias are and originate a highly positive impact to all present too. Next edition: 2022.


