[Free Full Text] Defective development and growth of the mandible in achondroplasia
An embryo is a new organism in the earliest stage of development. In humans this is defined as the developing organism from the fourth day after fertilization (when the union of a human egg and sperm cell occurs) to the end of the eighth week of pregnancy. After that, the unborn baby is usually referred to as the fetus.
Much of the embryonic developmental machinery (the cellular apparatus) used in human development is similar to that used by other animals. The machinery is essential for four processes: cell proliferation (multiplication), cell specialization (definition of which type of cell each cell will become), cell interaction and cell movement. During these processes approximately 20,000-25,000 genes in the human genome give rise to as many as 100,000 different proteins, which give the fetus form and substance.
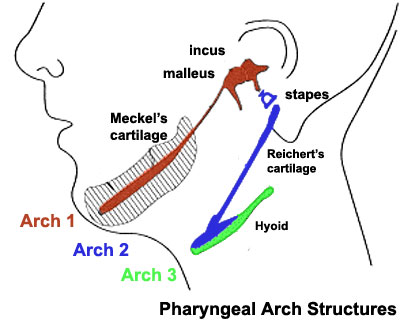
The pharyngeal arches (the same as branchial arch. "Branchial" comes from Greek and means gill) are a series of externally visible anterior (closer to the body) tissue bands lying under the early brain that give rise to the structures of the head and neck.
Each arch initially formed from similar components will differentiate (will be converted into other types of tissues) to form different head and neck structures. In humans, five arches are formed.
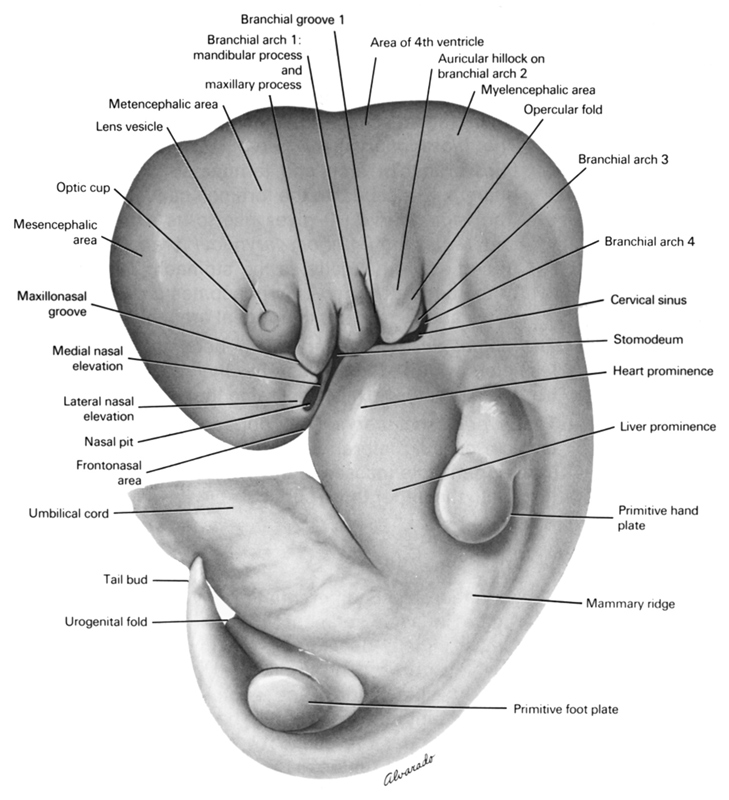
| Human embryo with 10 mm, with 5 weeks. Credits: The virtual Human Atlas. |
Arch 1 gives origin to a specific cartilage, designated Meckel's cartilage.
This introduction was made to frame the following study of Laurence Legeai-Mallet et al.: Meckel's and condylar cartilages anomalies in achondroplasia result in defective development and growth of the mandible, HMG Advance Access published June 3, 2016
"Mandibular growth relies greatly on primary and secondary cartilages. Meckel's cartilage (MC) is a rod-shaped primary cartilage that runs through the mandibular process of the first pharyngeal arch and acts as a template for the membranous ossification of the mandible body."
This study is sequential of the NVP-BGJ398 first study publication.
"Activating FGFR3 mutations in human result in achondroplasia (ACH), the most frequent form of dwarfism, where cartilages are severely disturbed causing defects on: long bones, cranial base and vertebrae. Since mandibular development and growth rely on cartilages that guide or directly participate to the ossification process, we investigated the impact of FGFR3 mutations on mandibular shape, size and position."
"we show that FGFR3 gain-of-function mutations lead to structural anomalies of primary (Meckel's) and secondary (condylar) cartilages of the mandible, resulting in mandibular hypoplasia and dysmorphogenesis."
"These defects are likely related to a defective chondrocyte proliferation and differentiation."
"The pan-FGFR tyrosine kinase inhibitor NVP-BGJ398 corrects Meckel's and condylar cartilages defects ex vivo. Moreover, we show that low dose of NVP-BGJ398 improves in vivo condyle growth and corrects dysmorphologies in Fgfr3Y367C/+ mice, suggesting that postnatal treatment with NVPBGJ398 mice might offer a new therapeutic strategy to improve mandible anomalies in ACH and others FGFR3-related disorder"
The main observations of this study were the following:
- Achondroplasia results in mandibular hypoplasia and dysmorphogenesis
- FGFR3 activation in mice results in mandibular hypoplasia and dysmorphogenesis
- Chondrocyte homeostasis is disturbed in Meckel's cartilage of Fgfr3Y367C/+ mice
- FGFR3 activation reduces condylar growth in humans and mice
- Tyrosine kinase inhibition corrects primary and secondary cartilages defects in Fgfr3Y367C/+ embryos
- Tyrosine kinase inhibition improves the mandibular dysmorphogenesis and the size of the condyle in Fgfr3Y367C/+ mice